Cooking with Woodfuels (Firewood and Charcoal)
Cooking Energy System | Basics | Policy Advice | Planning | Designing and Implementing ICS Supply | Designing and Implementing Woodfuel Supply | Climate Change | Extra
Overview
One of the characteristics of humankind is the ability to control fire and utilise it to prepare food. The oldest cooking fuel is firewood in the form of logs and branches from trees. Charcoal is the fuel derived from wood by pyrolysis in the process of carbonisation: the wood looses the volatile contents and the lighter yet energy-dense char remains. Due to the ease of transport and use, charcoal has become a prevalent cooking fuel in many urban areas of this planet, while firewood is more prevalent in rural areas.
Woodfuels
As firewood and charcoal have very different burning properties they need different devices (stoves) for cooking. This section deals with both fuel-types seperately.
- Cooking with Firewood
- Cooking with Charcoal
- Charcoal Cookstoves
- Dual Fuel Stoves
- Firewood Management Techniques
- Testing of Woodfuel Stoves
Efficient Cooking with Woodfuels
Wood fuels are the most common source of energy used for cooking. In the understanding of GIZ-HERA it comprises ‘firewood’ and ‘charcoal’. Firewood is wood from logs, sticks or twigs. Through carbonization it can be transformed into charcoal.
The use of woodfuels for cooking is commonly associated with deforestation and health problems. However, this is not an inherent problem of the fuel, but is strongly influenced by the quality and quantity of its usage. One possible solution is to improve the efficiency of the wood fuel usage.
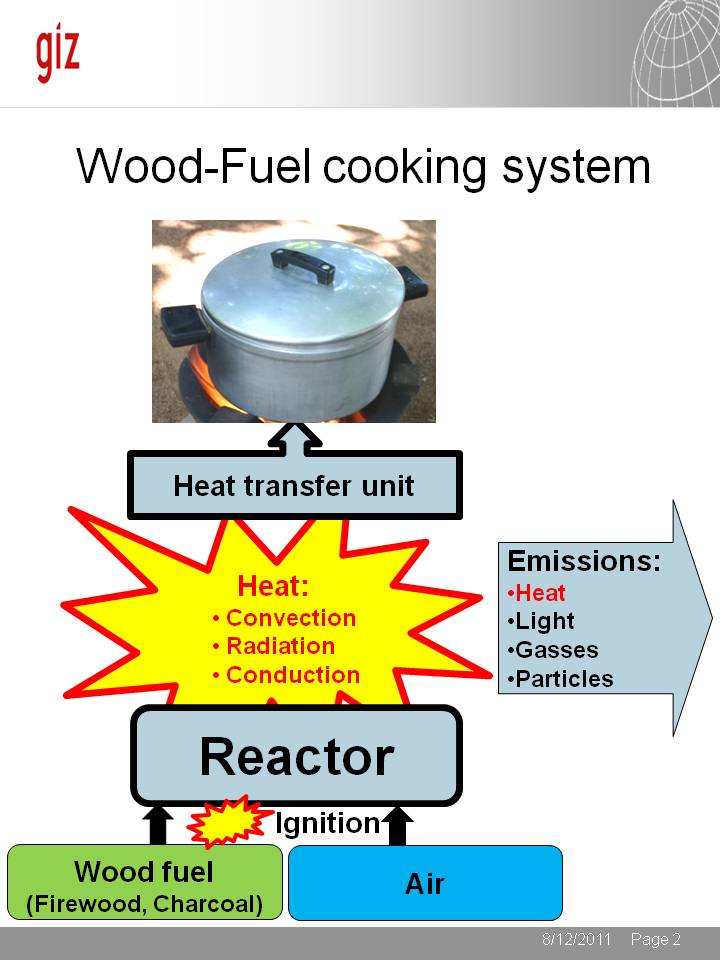
In the wood fuel cooking system, firewood or charcoal are mixed with air in a reactor. After ignition, a chain reaction is triggered in which heat is generated. This heat is transferred through 3 processes:
- Convection: Hot gasses are passing a surface transferring heat into surrounding materials
- Radiation: Red hot embers (charcoal) is radiating heat into surrounding materials
- Conduction: Heat is conducted through materials. Metal is a good heat conductor, whereas air is a poor heat conductor
The reactor is emitting heat, but also light, gasses and particles. While the emission of heat is wanted, the emission of gasses, particles and light are rather unintended. Good stove designs can reduce the quantity of unwanted emissions in favour of additional heat generation.
The heat does not enter automatically into a cooking pot. The design of the heat transfer unit has a big effect on the percentage of the heat transferred into the food to be cooked.
Overall there are two major dimensions for efficiency gains for wood fuel stoves:
- Achieve complete combustion (=‘create more heat per unit of fuel used’)
- Improve heat transfer (=‘get more heat actually into the pot’)
Firewood and charcoal are both wood-fuels. But charcoal is derived from carbonizing wood. In this process most volatiles are burned off. Hence the burning properties of firewood and charcoal are very different. While the dimensions for efficiency gains of all wood fuel stoves are the same, the solutions are different for firewood stoves as compared to charcoal stoves.
Wood-fire is a process in which woodgas is created and burned off. To accommodate these processes, firewood burning stoves require a high combustion chamber. Convection is the main form of heat transfer as hot flue gasses are created in the reactor and are passing the surface of the cooking pot. Hence the cooking pot sits above the flames with a good distance to the actual fuel. Fuel is continuously supplied and the heat is regulated according to the amount of fuel provided. Main emissions are smoke (particulate matter), light and to a lesser extent carbon monoxide.
In contrast, charcoal-fire is a process in which energy-dense charcoal is first heated to glowing embers (ignition phase). Oxygen is required to ‘burn’ the char to ash and gasses. There are far less emissions of particles in this process as compared to wood-fire. Heat is mainly transferred through radiation. This requires that the cooking pot sits close to the surface of the fuel, making the combustion chamber rather shallow. Charcoal stoves are batch-fed (all fuel required is filled in a container and lit at the start of the cooking process) and air-regulated: more air, more heat. For simmering, heat can be reduced by lowering the input of air to the reactor. Carbon monoxide is the main emission, whereas light and smoke (mostly during preheating) play a minor role.
Further Information
References
This article was originally published by GIZ HERA. It is basically based on experiences, lessons learned and information gathered by GIZ cook stove projects. You can find more information about the authors and experts of the original “Cooking Energy Compendium” in the Imprint.